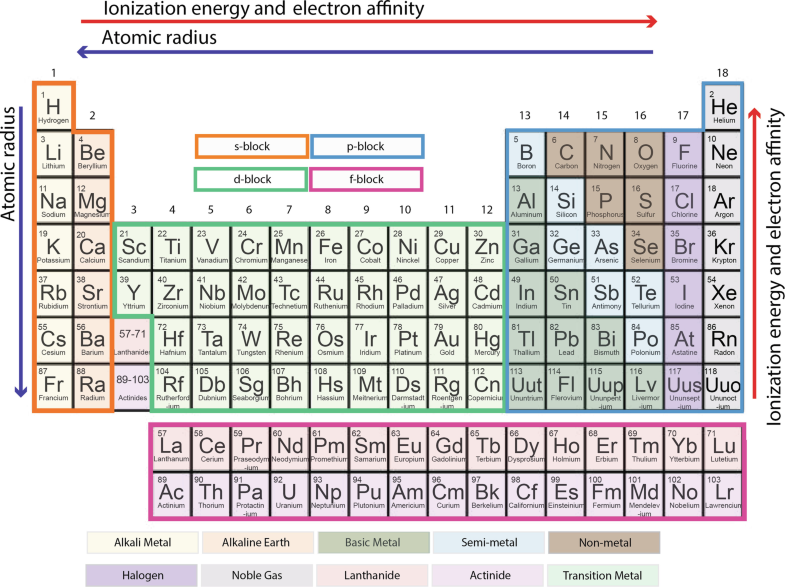
They are metals Unpredictable reactivity and propertiesħ CLASSES OF ELEMENTS Lanthanides – atomic number 57 – 70Ī. The atomic mass in grams is extremely small, so scientists developed a unit based on the carbon atom Atomic mass unit (amu) – equal to 1/12 the mass of a carbon-12 atomĥ CLASSES OF ELEMENTS Metals – left and center part of periodic tableĬonduct heat and electricity, have luster, malleable, ductile All are solid at room temperature (except mercury)Ħ CLASSES OF ELEMENTS Transition elements – groups 3 – 12 Period – horizontal row Group – vertical columns they are numbered 1 – 18 Family – specific name describing one or more groups ex: Column 18 is the noble gases Elements in the same group have the same number of valence electrons (electrons in their outer shell)Ĥ Atomic Mass Atomic mass depends on the distribution of an element’s isotopes in nature and the masses of those isotopes. Since the valence shell is three, the period will also be 3.2 The Periodic Law In the modern periodic table, elements are arranged by increasing atomic number (number of protons) Properties of elements repeat in a predictable way when atomic numbers are used to arrange elements into groups. Hence the remaining 7 will be in the third shell as the third shell has a capacity of 18 electrons. The first shell has 2 electrons and the second will have 8. Q: Electronic configuration of element T is 2, 8, 7. Learn more about D Block and F Block elements here in detail here.

The number of electrons in this last shell increase by one across any given period All the elements in the period have the same number of shells.
:max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
There are seven such periods in the table, each numbered from 1 to 7.Horizontal rows of the periodic table of elements are the periods.Earlier Attempts at the Classification of Elements.Browse more Topics under Periodic Classification Of Elements Now, this matrix divides itself into rows and columns. There are 118 elements in total, spread out on the periodic table. The arrangement of these elements is in a grid or matrix format. So the periodic table of elements has been arranged in increasing order of their atomic numbers. “Properties of elements are a periodic function of their atomic number” The arrangement of elements in the periodic table follows a certain rule known as the periodic law, which is:

